SERVICES
Solvent Recovery

ReCoveRY oF
soLvents AnD CHemICALs
The demands for recovery of useful solvents and other chemicals
from the waste continue to increase in order to meet environmental
regulations and reuse recovered solvents and chemicals to reduce
operating cost. TPT can provide innovative solutions to waste
treatment problems using the advanced separation technologies;
Distillation/Azeotropic Distillation
Extractive Distillation
Liquid-Liquid Extraction
Extraction/Distillation Systems
Absorber/Stripper Systems
Adsorption
APPLICATIONS:
Recovery of solvents from a large-volume of vent gas containing
thin solvents
Recovery of Carboxylic Acids from waste water formed in the
DMT, PTA, paper and pulp, and other chemical plants
Aromatic recovered from refinery and petrochemical plants
Useful Gas recovered from vent gas
Alcohols recovered from waste water
Primary, Secondary & Tertiary Buthyl Alcohol and Ethanol,
Methanol, IPA, Butanol etc…
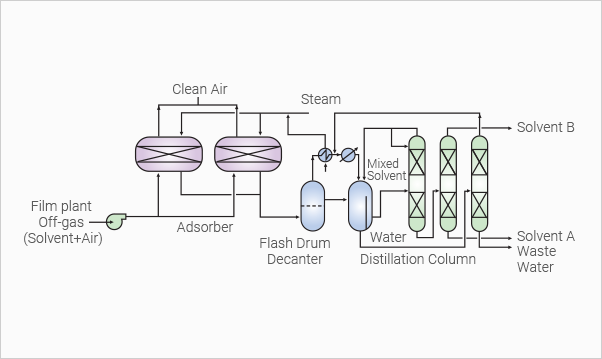
Solvent Recovery
From Waste Gas
TPT’s solvent recovery process is based on an activated carbon
adsorption method which recovers solvents from a large-volume
of vent gas containing thin solvents This process treats such
solvents as THF, Ketone and Anone without degrading or
polymerizing. The recovered solvent is perfectly reclaimed,
and identical to fresh solvent. Costs for solvents can be
remarkably reduced, and at the same time, pollution control
can be achieved completely.
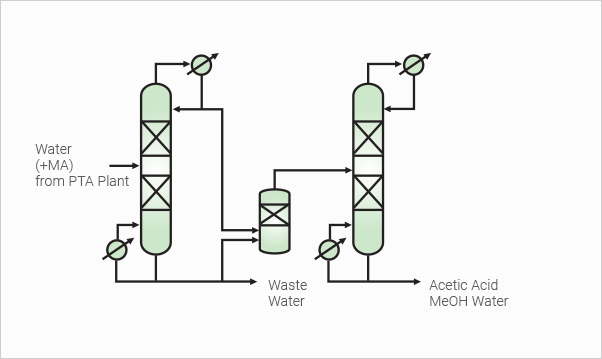
Methyl Acetate Hydrolysis Plant
Methyl Acetate (MA) is generated during oxidation of PX with
solvent(Acetic Acid) in PTA plant. MA can be recovered by
concentrating of waste water and this MA is raw material of
this plant. TPT’s MA Hydrolysis process produces Acetic Acid,
Methanol and water mixture with high yield and efficiency. Acetic
Acid & MeOH separation tower can be added by customer’s need.
This process has already been proven in commercial operation
and is getting good reputation from PTA makers who get
MA as a by product.
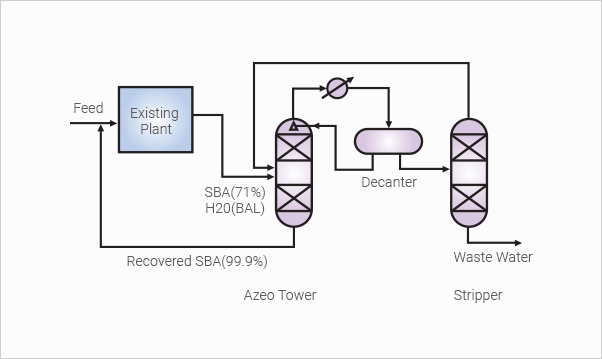
Sec-Buthyl Alcohol
(SBA) Recovery
Highly nonideal solutions like this SBA and water mixture can
be separated by azeotropic distillation technology with entrainers.

- A.
- 1st Floor Hanaro Building, 25, Insadong 5-gil,
Jongno-gu, Seoul, 03162, Korea (Head Office)
- T.
- +82) 2-6356-2550 (Head Office)
- T.
- +82) 52-237-2720 (Shop)
- E.
- tpt@tptpacific.com


